Third-Party Manufacturers
We will provide a golden opportunity for renowned people who want to take advantage of third party manufacturing services.
We will deliver quality medicines at reasonably priced and wholesale prices and are highly situated as a leading third party manufacturing pharmaceutical company. We invite any direct manufacturers company to walk with us as we offer a wide range of business ideas.
Third--party manufacturing pharmaceutical (our role)
Our company is among the leading third party manufacturing pharmaceutical companies in India. With the help of our large-scale production unit, we have been able to offer our associates a wide variety of pharmaceutical products. Being one of the most reputable third party pharmaceutical manufacturers, we have the ability to provide all kinds of pharmaceuticals such as pills, tablets, capsules, herbal nutraceuticals, protein powders, gym supplements, capsules, pharmaceutical tablets and sachets. Being a leading third-party manufacturing pharmaceutical company, we use 100% best quality raw material and state-of-the-art equipment.
Below are some of the pharmaceutical manufacturing services we offer. Let's take a look –
Here given some important our product range available :- Tablet Manufacturing
- Capsule Manufacturing
- Syrup Manufacturing
- Softgel Manufacturing
- Spray Manufacturing
We are linked with the best distribution channels in India, which help us to make the on-time delivery of all the manufactured products. Our domain lies in the list of the top 10 Third Party Pharma, allopathic, and Natural medicine manufacturers in India. Along with the quality of drugs, we also ensure the quality packaging of all the produced drugs. Being the top Third Party Pharma Manufacturer in India currently, we will provide 100% genuine services. We are providing our service to around 350+ pharma companies from all around the nation.
About Third Party Manufacturing & Contract Manufacturing Facilities
Product Approvals – We Have Both Complete Antibiotic Range & Non-Beta Lactam Manufacturing Approvals.
We Provide All Services Regarding Manufacturing Like, Private Label Manufacturing, Custom Formulation Manufacturing, and government-level Manufacturer.
Product Options Available in Manufacturing Plant/Unit with Production Capacity: Here given -:
| Tablet | 3.5 Billion Units Tablets (per annum) |
| Syrup | 10.2 million liters Liquid (Per Annum) |
| Capsules | 190.5 Million Units Capsules (per annum) |
| Drops | Millions Units Drops (Per Annum) |
| Inhalers | Millions of Units Inhalers (Per Annum) |
| Sachet | Millions Units Sachet (Per Annum) |
| Powder | Millions Units Powder (Per Annum) |
| Soft Gel | 145.2 Million Units Capsules (Per Annum) |
| Gel | Millions Units Gel (Per Annum) |
| Spray | Millions of Units Spray (Per Annum) |
| IV Fluid | Millions Units IV Fluid (Per Annum) |
| Nasal Spray | Millions of Units Nasal Spray (Per Annum) |
| Majoon | Millions of Units Mazoon Spray (Per Annum) |
| Khamira | Millions of Units Khamira (Per Annum) |
| Liquid | Millions of Units of liquid (Per Annum) |
| Creams | Millions of Units of cream (Per Annum) |
| Oil | Millions of Units of oil (Per Annum) |
Product Sections Available In Manufacturing Plants/Unit with Production Capacity :-
Our Certification
Our Certification
Our product is manufactured with international standards; the plant has all the necessary certifications to meet international standards.
Documents Required for 3rd Party Manufacturing
- 1 Profile
- 2 Information
- 3 Copy of Resolution
- 4 Drug Licenses
- 5 GST
- 6 Agreement
- 7 Certificate
Profile:
(Brief Profile with Copy of PAN Card of the company and a Copy of Memorandum & Articles of Association in case of Pvt. Ltd or limited Company. Partnership deed/ Affidavit for proprietary in case of Partnership firm or Proprietorship firm.)
Name, Address & Telephones with copy of Aadhar Card and Pan Cards.
(Of all Directors, Partners or Proprietor both official and residential.)
Copy of Resolution for Authorized Signatory to Deal
(For limited, private limited and partnership companies)
Drug Licenses:
(Attested copy of Drugs Licenses to be provided)
GST Registration Certificates:
(Attested copy of Sales tax Registration Certificate to be provided)
Agreement for Manufacturing:
(Specimen Copy Will Provide)
Certificate for Non-Resemblance:
(Specimen Copy Will Provide)
Process
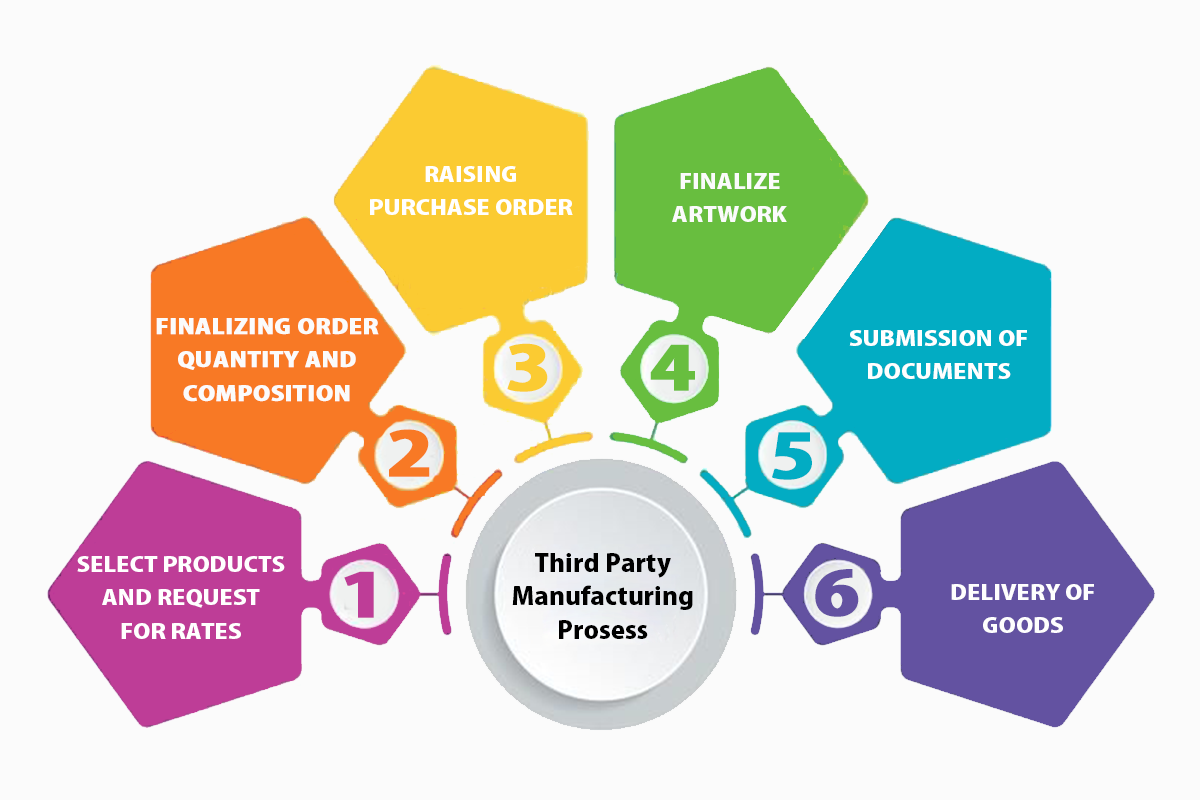
PROCESS OF THIRD PARTY CONTRACT MANUFACTURING
Third-Party Manufacturing business in the pharma industry is growing at a very fast pace globally and is expected to increase at a very fast rate over the next few years. Every manufacturing business company is using quite similar technologies and equipment.
The third-party manufacturing process consists of multiple steps, and for each step, there are process parameters with some control limits.
- Finalizing order quantity and composition
- Raising quotation
- Required documents
- Packaging material
- Product manufacturing
- Product delivery
- Big-level products import and export
Expected Investment
Expected Investment Required for Allopathic Or Ayurvedic Third Party Products Manufacturing Process
What Does Inventory Require to Get Manufactured Pharma / Ayurvedic product at Third Party Basis?
Inventory may vary according to your product selection, dosage form, packing material requirement etc. Your inventory also may vary according to batch size of product. Less expensive product will require less investment but their batch size may large in quantity. Likewise costly products require more inventory but their batch size may be small in quantity. Some manufacturer take small batch for all products, hence one have to invest less. There is not a fixed amount of investment for getting manufactured your product at third party basis. Roughly you can calculate your investment according to below given information’s
Tablet/Capsule
Batch Size: 25000 tab/cap
Pack: 10*10 Blister/alu-alu
- Packing material require: 250 Box and roughly 2-3 kg foil. But printer will not print such small quantity. You have to print minimum 500 or 1000 boxes and 5 kg of foil. One time 1100/- cylinder charges for one color and 2200/- for two color foil cylinder charges, you have to pay to printer.
- Estimated cost will be calculated as per 1000 box printing material:
- Box= 1000*1.5=1500
- Foil=5*450=2250
- Cylinder charges: 1500
- Total packing material inventory: 5250/-
- Total number of box to be manufactured is 250*90=22500
- Packing material inventory= 5250
- Total cost= 27750/-
- Now calculate the costing for first batch: 27750/250=111/- per box.
- Now calculate the costing for next batch: 22500/250=90/- For next 2 batch, you may not require new box printing but you may require some foil. Here we are calculating costing for first two batches only.
- Average costing= 111+90= 201/2= 100.5/- per box.
Ointments/Creams/Gel
Batch Size: 1000 pc
Pack: 20 gm with outer cartoon
- Packing material required is 1000 pc
- Box: 2000 pc*1/-= 2000/-
- Lami tube: 10000 pc*2/-=20000/-
- Cylinder charges for one color is 1500/- and for two color 2500/-(one time investment)
- Total Packing material: 23500/-
- Manufacturing cost: 6.5*1000= 6500/-
- Packing material inventory: 23500/-
- Now calculate the costing for first batch: 30000/1000=30/- Per pc
- Now calculate the costing for next batch: 6500/1000=6.5/-
- After next 10 batches, you don’t need to invest in Lami tube. But after 2000 pc, you will need boxes for this and you can add 1/- per pc more costing.
- Now costing for next 10 batches costing will be 7.5/- per pc
- Average costing: 30+6.5+7.5+7.5+7.5+7.5+7.5+7.5+7.5+7.5=96.5/10=9.65/- per pc.
Liquid/Syrup
Batch Size: 1000 pc
Pack: 60 ml. with outer cartoon
- Box: 2000 pc*0.75/-= 1500/-
- Label: 5000 pc*0.10/-=500/-
- Total Packing material: 2000/-
- Manufacturing cost: 6.5*1000= 6500/-
- Packing material inventory: 2000/-
- Now calculate the costing for first batch: 8500/1000=8.5/- Per pc
- Now calculate the costing for next batch: 6500/1000=6.5/-
- Average costing: 8.5+6.5=15/2=7.5/- per pc
Injections
Batch Size: 2000 pc
Pack: 1 ml with outer cartoon
- Packing material required is 5000 pc
- Box: 5000 pc*0.40/-= 2000/-
- Label: 5000 pc*0.05/-=250/-
- Total Packing material: 2250/-
- Manufacturing cost: 3*2000= 6000/-
- Packing material inventory: 2500/-
- Now calculate the costing for first batch: 8500/2000=4.25/- Per pc
- Now calculate the costing for next batch: 6000/2000=3/-
- Average costing: 4.25+3=7.25/2=3.63/- per pc
Above costing is for example purpose only. It is not the actual cost sheet of any product. According to above basis, you can calculate, how much you have to invest initially to start you own pharma marketing company.
Product manufactured from Leading Pharma Manufacturing Units





F.A.Q
Frequently Asked Questions
-
What is the importance of the Pharmaceutical Contract Manufacturing Organization (CMO)?
A contract manufacturing organizations (CMO) or private label manufacturing is an organization that serves the pharmaceutical industry and provides customers with a wide range of services, from drug development to manufacturing. Outsourcing management to a CMO allows pharmaceutical clients to expand their technical resources without overhead.
-
What is the importance of third-party manufacturing in the pharmaceutical industry?
According to trade theory, third-party manufacturing is a manufacturing process for third-party drugs and products. In this process, an agreement is signed between the company and the manufacturing company, and the manufacturing company manufactures the products according to the contract.
-
What is the difference between contract manufacturing and third party manufacturing?
This is very important; in third party manufacturing there is no time limit for delivery of goods, while in contract manufacturing there is a time limit for delivery. Most of the small companies work on the basis of third-party manufacturing, while the medium and large companies work on the basis of contract manufacturing.
-
What is the procurement process, inventory and types of packaging material in pharmaceutical/Ayurvedic manufacturing companies? Or What is the packaging material for pharmaceutical/ayurvedic products?
In our TPM, the packaging material is the outer covering in which the dosage form is packaged. It can be made of paper, plastic, rubber, tin or any other form. Packaging materials include outside cartoon, monkey cartoon, aluminum foil, label, chimney, etc. Vials, ampoules, protein powder cans, and PET bottles are generally not considered packaging materials, but they are also part of packaging materials. It is considered part of the product. The manufacturer will give you the rates that include bottles, vials, etc. even if you request the rates without packing material. In the pharmaceutical industry, there is a standard that must be printed as packaging material. It is very important to keep this in mind.
Types of Packing Material:
- Box
- Foil
- Label
- Lami Tube
- Inner/mono ( if applicable)
- Large or small cartoons (plastic, wood, aluminum and others)
How to ensure product quality in third party/contract manufacturing?Query:
We were planing to start pharmaceutical marketing company, read your posts. we found it very helpful. Still we have one confusion; if drug product manufactured by third party shows some adverse reactions then in that case DCGI take action against whom marketing or manufacturing company? and what will be the penalty for accused?
Response:
Manufacturer will manufacture only DCGI approved molecules. These molecules and combinations are already tested toward adverse reactions. In some cases if products show any adverse reactions or drug department found product of substandard quality then manufacturer will be responsible for it. Marketing company has to show proof of sale and purchase in that case and satisfy the drug department regarding marketing company don’t have any knowledge about substandard quality of product.
But its also responsibility of marketing company to take quality report along with product or test each batch at independent laboratory for quality and efficacy of product.
DCGI can make accused manufacturing company or in some cases it make accuse all distribution channel along with marketing company. Penalty could be only fine or imprisonment or both depend at severity of matter.
Query:
I’d like to ask you one more question. Though manufacturing company has already got the approval for drug product from DCGI. But, what if they produce drug products which are not having API strength specified or they provide products with 100% assay value but product not remain stable during shelf life and undergo degradation, hence reduction in assay value. As a marketing company we are planning for contract manufacturing, so in that case how can we ensure quality of products? Can you please reply on this? I would be really grateful if you could answer.
Response:
In case of product that are not produced with quality standard by manufacturer or not having API strength as specified. You have two option:
First ask COA (Certificate of Analysis) along with Batch you received from manufacturer.
Second send few sample to independent laboratories for testing. Many laboratories provide facility for testing products and will give you assay report of batch you received from manufacturer.
What is the Minimum Order Quantity (MOQ) for third party manufacturing?It Depends Upon Company As Well As Product Section , Its Varies Company To Company , Party Can Negotiate With Manufacturing Company .
Why do third party/Contract Manufacturer require Trade Name/ Brand Name Affidavit?Query:
Requesting your help regarding drug brand name registration I run a proprietorship company in Bangalore pcd business already but want to enter contract manufacturing. Companies are demanding brand Name Affidavits. I just want to clarify that
Response:
Companies demand brand name Affidavit for avoiding any future problem. In Pharma daily new molecule launch and companies has to search for a unique Trade Name/Brand Name for it. One most common way to search unique brand name to search at IP INDIA website and know whether your brand name is resembling to any other or not. But there is lot of brands available which are not applied for brand name registration. That can’t be searched by it. So, there is maximum chances, any new chosen trade name could clash with any already existing trade name.
Manufacturing companies will ask for brand name affidavit because they want to confirm that you are not using any other company brand name. You Brand Name/Trade Name is unique and don’t resemble to any other Brand Name/trade Name available in Market. If in Future your Brand Name will clash with any other Company’s Brand Name, you will have total responsibility for it. Manufacturing company will not bear any responsibility and expenditure with regard to this brand name.
If one company is marketing by Name of Sovamax, you can’t choose any Trade Name/Brand Name Similar or resembling to it like Aovamax. It will be offense under Intellectual Property Act. You have to choose different Name like Kdakmax etc. For entering into contract manufacturing, you should set-up your new marketing company and have to choose brand names for it.
Contact
Contact Us
Our Address
Plot No. 1770, HSIIDC, Rai Industrial Estate Phase, 1, Sector 38, Sonipat, Haryana 131029, India
Email Us
Call Us
+91-8684845757















 Call us
Call us Whatsapp
Whatsapp Contact us
Contact us